|
7/6/2023 0 Comments Praxbind cost
Some of these patents protect the original drug, whereas others may protect follow-on inventions or even inventions casually mentioning the drug. The expiration dates listed for these patents are estimates, based on the grant date of the patent.įor completeness, these patents were identified by searching the patent literature for mentions of the branded or ingredient name of the drug. As a guide, Praxbind intravenous solution (2.5 g/50 mL) usually costs 4,700 for. 
This list may exclude some of the patents which block biosimilar launch, and some of these patents listed may not actually block biosimilar launch. Praxbind cost without insurance will vary depending on where you buy it. These patents were identified from searching drug labels and other general disclosures from the brand-side company. The expiration dates listed are not estimates - they're expiration dates as indicated by the brand-side company. Currently Praxbind is available in SA as a section 21 drug at a cost of. They have a high certainty of blocking biosimilar entry. Rates of major bleeding for dabigatran 150 mg and dose-adjusted warfarin appear. These patents were identified from disclosures by the brand-side company, in response toĪ potential biosimilar seeking to launch. price for the recommended 5 g dose of idarucizumab is 3500. 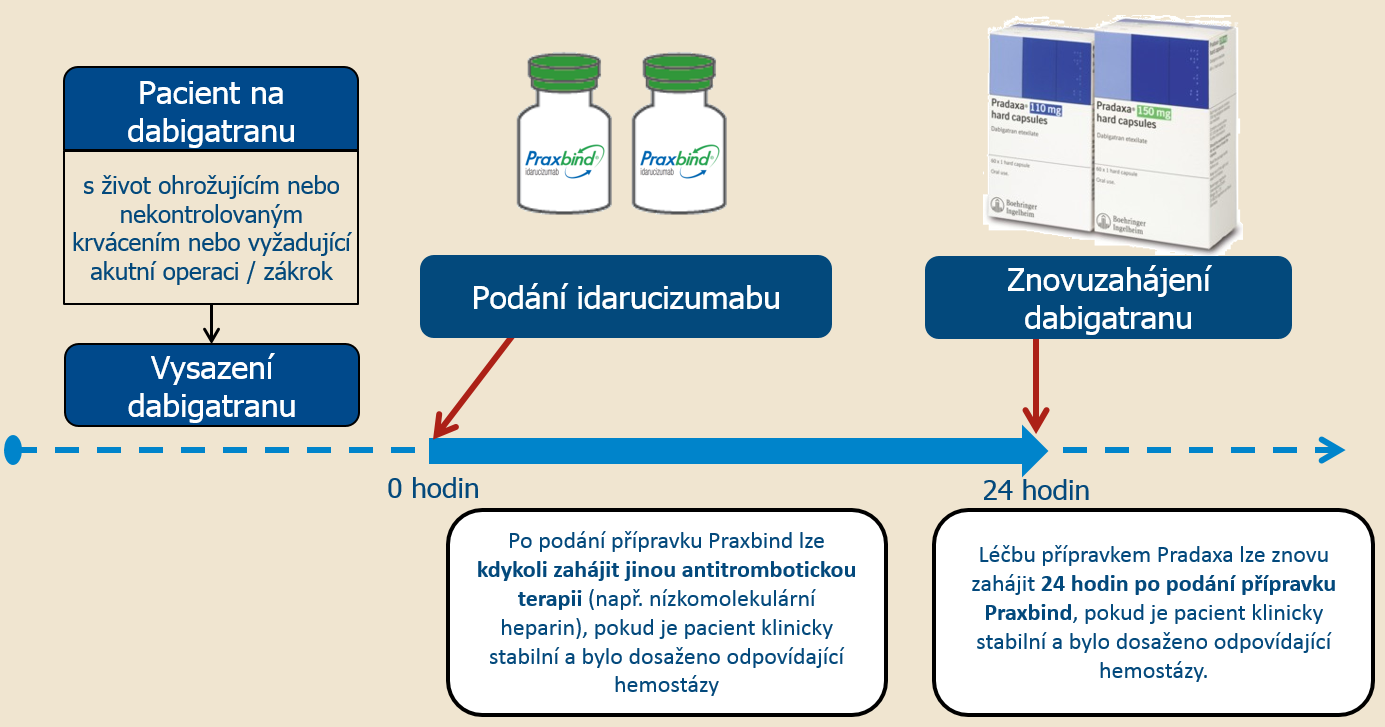
It proved effective in lab trials, but is it ready for prime time in the ED. Hospitals can alleviate cost burden by obtaining DOAC reversal agents on consignment through their respective vendor or, if not available on consignment, keeping a limited number of doses on hand based on. On October 16th, the FDA approved the use of idarucizumab (Praxbind®), a monoclonal antibody developed by Boehringer Ingelheim Pharmaceuticals for the reversal of dabigatran. Prices are for cash paying customers only and are not valid with insurance plans.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
